Sleep Disturbance in Perimenopause: An Early, Evidence-Based Health Signal
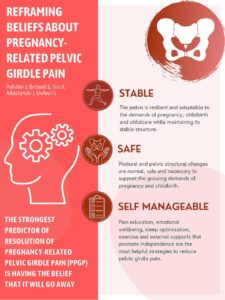
Sleep disturbance is one of the most common and distressing concerns reported by women during the perimenopausal transition. Importantly, growing evidence shows that disrupted sleep often begins years before the final menstrual period, and cannot be explained solely by ageing or vasomotor symptoms such as hot flushes.
The Study of Women’s Health Across the Nation (SWAN) has provided original insights, more recent high-quality research confirms that perimenopausal sleep disturbance is an early, biologically driven phenomenon, linked to hormonal variability and interacting physiological systems.
What the research shows
Longitudinal and mechanistic studies demonstrate that during perimenopause, women experience increased night-time awakenings, fragmented sleep, difficulty maintaining sleep, and non-restorative sleep, even when total sleep duration appears unchanged (Baker et al., 2022). These sleep disruptions have been shown to correlate with rising follicle-stimulating hormone (FSH) and fluctuating oestradiol levels, and notably can occur independent of hot flushes or night sweats.
Meta-analytic data further confirm that sleep disturbance during perimenopause is multifactorial, influenced not only by hormonal changes, but also by mood symptoms, anxiety, metabolic health, and emerging cardiometabolic risk factors (Zhang et al., 2023). This aligns with newer community-based studies identifying distinct sleep-disturbance profiles in perimenopausal women, including difficulties with sleep initiation, sleep maintenance, and early morning waking, each associated with different physiological and psychosocial contributors (Li et al., 2025).
Why sleep changes occur before menopause
Perimenopause is characterised less by absolute oestrogen deficiency and more by hormonal variability and disrupted signalling. Oestradiol plays a role in sleep regulation through its effects on thermoregulation, circadian rhythms, neurotransmitters, and vascular function. Fluctuating levels — rather than low levels alone — appear to destabilise these systems, contributing to sleep fragmentation well before menstruation ceases.
Emerging evidence also links perimenopausal sleep disturbance with early endothelial and cardiometabolic changes, suggesting that poor sleep may be both a symptom and a contributor to broader health risk during the menopause transition (Khoudary et al., 2020; Davis, 2024).
Clinical relevance
From a clinical perspective, this body of evidence reframes sleep disturbance in perimenopause as a meaningful health signal, not simply a nuisance symptom to be endured. Poor sleep during this transition is associated with reduced exercise tolerance, impaired recovery, mood disturbance, increased visceral fat accumulation, insulin resistance, and rising cardiovascular risk.
Recognising sleep disruption early provides an opportunity for preventative, whole-system care, including education, lifestyle and exercise interventions, psychological support, and — where appropriate — discussion of evidence-based menopausal hormone therapy options within an individualised risk–benefit framework.
Key takeaway
Current evidence clearly shows that sleep disturbance is a core feature of perimenopause, emerging early in the transition and reflecting complex interactions between hormonal variability, vascular health, metabolic changes, and psychosocial stressors. Addressing sleep proactively is therefore central to supporting women’s health, function, and long-term wellbeing during midlife.
References (APA 7th)
Baker, F. C., de Zambotti, M., Colrain, I. M., & Bei, B. (2022). Sleep problems during the menopausal transition: Prevalence, mechanisms, and consequences. Journal of Clinical Endocrinology & Metabolism, 107(10), e4144–e4156. https://doi.org/10.1210/clinem/dgac423
Khoudary, S. R. E., et al. (2020). Menopause transition and cardiovascular disease risk: Implications for timing of early prevention. Circulation, 142(25), 2403–2414. https://doi.org/10.1161/CIRCULATIONAHA.120.047182
Li, X., et al. (2025). Sleep quality profiles and associated factors in perimenopausal women: A community-based study. BMC Women’s Health, 25, Article 217.
Zhang, Y., et al. (2023). Risk factors for sleep disorders in perimenopausal women: A systematic review and meta-analysis. Menopause, 30(9), 1021–1032.
Sleep Changes in Perimenopause: What This Means for You
Many women notice changes in their sleep years before their periods stop. This can include waking during the night, lighter or less refreshing sleep, difficulty staying asleep, or feeling tired despite “getting enough hours.”
Importantly, research now shows that these sleep changes are not just about hot flushes or getting older. During perimenopause, hormones such as oestrogen don’t simply drop — they fluctuate. This hormonal variability can affect how the brain regulates sleep, body temperature, mood, and stress responses.
Sleep disruption in this stage is often part of wider body changes happening at the same time. These may include:
- increased fatigue or reduced exercise tolerance
- changes in mood or anxiety
- weight gain around the abdomen
- blood pressure or blood sugar changes
This tells us that poor sleep during perimenopause is a signal, not a failure — a sign that the body is adapting to a major transition.
A whole-person approach matters
Addressing sleep in perimenopause works best when we look beyond a single hormone or symptom. Support may include:
- education about what’s happening in the body
- movement and strength training that supports nervous system regulation
- strategies to support recovery and energy
- discussion of evidence-based medical options when appropriate
He wā whakawhiti tēnei — this is a time of transition.
Supporting sleep early can help protect long-term wellbeing, energy, and cardiovascular health.
The key message
If your sleep has changed in your 40s or early 50s, you’re not imagining it — and you’re not alone. These changes are common, real, and increasingly well-understood. With the right support, sleep and wellbeing can improve.
Ko te moe te pou o te oranga — sleep is a pillar of health.
If sleep disruption is affecting your energy, recovery, or quality of life, early support can make a meaningful difference.
References:
Baker, F. C., de Zambotti, M., Colrain, I. M., & Bei, B. (2022). Sleep problems during the menopausal transition: Prevalence, mechanisms, and consequences. Journal of Clinical Endocrinology & Metabolism, 107(10), e4144–e4156. https://doi.org/10.1210/clinem/dgac423
El Khoudary, S. R., Aggarwal, B., Beckie, T. M., et al. (2020). Menopause transition and cardiovascular disease risk: Implications for timing of early prevention. Circulation, 142(25), 2403–2414. https://doi.org/10.1161/CIRCULATIONAHA.120.047182
Li, X., Zhang, Y., Wang, Y., et al. (2025). Sleep quality profiles and associated factors in perimenopausal women: A community-based study. BMC Women’s Health, 25, Article 217.
Zhang, Y., Liu, H., Li, S., et al. (2023). Risk factors for sleep disorders in perimenopausal women: A systematic review and meta-analysis. Menopause, 30(9), 1021–1032.
Davis, S. R. (2024). Understanding cardiometabolic and vascular changes across the menopause transition [Paul Dudley White International Lecture].